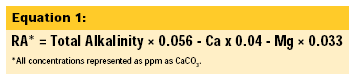Importance of water source
Long before the water chemistry behind the brewing process was scientifically understood, great breweries sprang up in the proximity of excellent water sources. If the local water source was inadequate for proper brewing, the beer produced would be of poor quality. Either the brewery moved to a better source of water, adapted their brewing techniques, or they perished.
Water profiles from the classic brewing regions are surprisingly different from each other, accounting for the evolution of unique beer styles from those areas (see www.barandbeer.com or www.ebraumeister.com for beer types). It was the early trial and error compensating methods in addition to the presence of certain mineral ions that determined the style of beer that evolved from an area. When certain Munich brewers moved to Prague, they were amazed at how light and delicate their beers turned out from essentially the same recipes and ingredients used in Munich.
What's most important to the brewer is the effect the brewing water has on mash pH. Malt enzymes are very particular about temperature and pH, and mashing is essentially an enzymatic process. Proper mash pH, usually preferred at around 5.2-5.4, can be achieved by several means. In the early days of brewing, methods were mostly limited to malt composition and biological acidification. Modern breweries may add food grade acids such as lactic acid or phosphoric acid to adjust the mash pH.
London water is high in carbonate ions, which tends to drive the mash pH up, but dark roasted malts acidify the mash and the balanced pH results in excellent porters. North of London in an area called Burton Upon Trent the water has extremely high levels of dissolved minerals, especially gypsum, which lowers mash pH, thus enabling a balanced mash with minimal dark malts resulting in exceptional pale ales.
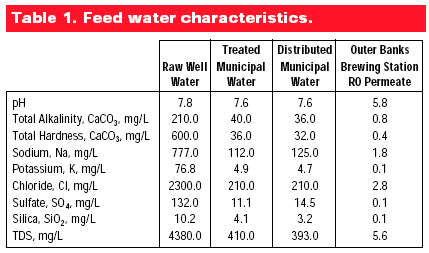
It's interesting to note here that both Prague and Burton Upon Trent produce world class light beers while Prague has very low total dissolved solids (TDS) - around 31 parts per million (ppm) - and Burton has exceptionally high TDS (around 1,226 ppm). The reason they both work for brewing is the overall balance of ions and especially the residual alkalinity (RA):
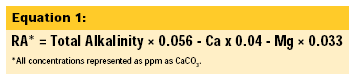
RA values less than 1 are considered good brewing water. By this formula, Burton Upon Trent water is actually superior to Pilsner (Prague) water. In fact, the practice of adding gypsum to brewing water to achieve a lower mash pH is still sometimes referred to as "Burtonization."
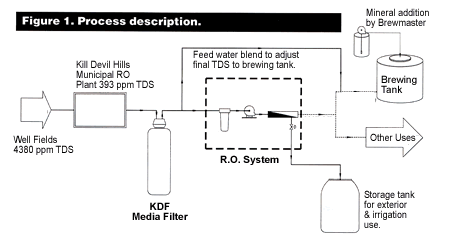
There are many subtleties to managing the ion balance. While it's essential to keep the mash in the right pH range by whatever means, the presence of certain minerals does have an effect on flavor and character of the beer. Many minerals have a positive effect on brewing up to a point, after which they're detrimental, causing excessive saltiness, sourness or bitterness. Yeast requires a proper balance of calcium (Ca) with magnesium (Mg) for optimal fermentation along with several other nutritional requirements. Thus, absolutely pure water would not make very good brewing water. But by using RO as pretreatment for brewing water, the brewer can add back minerals to suit any style of beer he or she is trying to create or duplicate.
 Microbrewery Application
Microbrewery Application